
IVD laboratory workflow kits
Analyzer-adjacent consumables, validation binders, and LIS handoff checklists for laboratories that need clear purchasing records.
CLIA filesHL7 notesQC ready
Standardize dental equipment, IVD workflow tools, and infection-control consumables through one documentation-ready commercial channel.
Envista separates routine replenishment from validation-heavy equipment programs so buyers can move quickly without losing audit evidence.
| Requirement | Dental Equipment Program | Lab & Infection Control Program |
|---|---|---|
| Core documentation | UDI-DI sheet, installation checklist, preventive maintenance plan | Lot file, IFU library, cold-chain or storage statement |
| Integration notes | DICOM, chairside imaging export, service calibration record | HL7 v2.5.1, LIS handoff, reagent stability controls |
| Buyer workflow | Capital request, site readiness, biomedical acceptance | Standing order, par-level replenishment, variance review |
| Service target | 48-hour remote triage with escalation for installed base issues | Same-day documentation response for batch and recall inquiries |
| Commercial file | Quote, accessory matrix, warranty terms, training slot | SKU crosswalk, MOQ, shelf-life, packaging configuration |

Analyzer-adjacent consumables, validation binders, and LIS handoff checklists for laboratories that need clear purchasing records.
CLIA filesHL7 notesQC ready
Lot-traceable supply bundles for sterile processing, dental operatories, and clinical storage teams maintaining predictable par levels.
Lot traceIFU accessRecall flow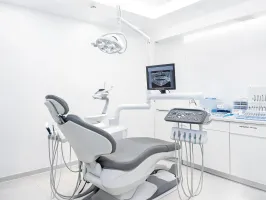
Equipment packages for DSOs and clinics that require install plans, accessory matrices, and concise service expectations.
Install planPM scheduleTraining file
Every product family is framed around ordering identifiers, compatibility notes, service expectations, and evidence references. That keeps value-analysis review lean and reduces back-and-forth before purchase orders are released.

Envista service files distinguish remote triage, replacement workflows, and preventive maintenance intervals, so distributed clinics and laboratory sites know what happens after the first shipment.